|
With initial electrophilic attackĪddition reactions beginning with electrophilic attack include many additions to olefins (compounds with double bonds), some additions to acetylenes ( compounds with triple bonds), and some additions to compounds with other multiple bonds. Many mechanisms are known for such reactions most of them are variants of four basic mechanisms, which differ chiefly in the sequence of events that occur. Reactions in which a multiple bond between two atoms becomes partly or fully saturated by covalent attachments at both centres are called addition reactions. Examples of these reaction sequences are shown below: Addition reactions An example is shown below in which the positions of chlorine attachment and proton loss were established by isotopic labeling.Īddition-elimination and indirect substitution reactions also can occur and are responsible for a number of unusual products formed in aromatic substitution reactions. Substitution with accompanying rearrangement of the double-bond system is another established reaction path. 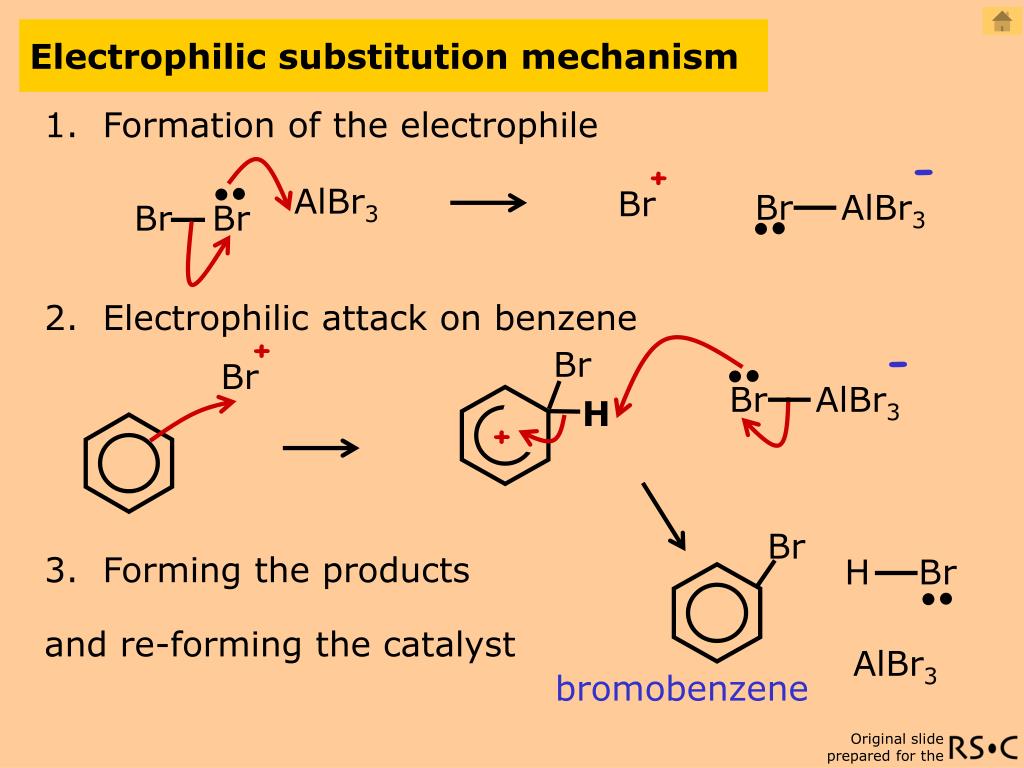
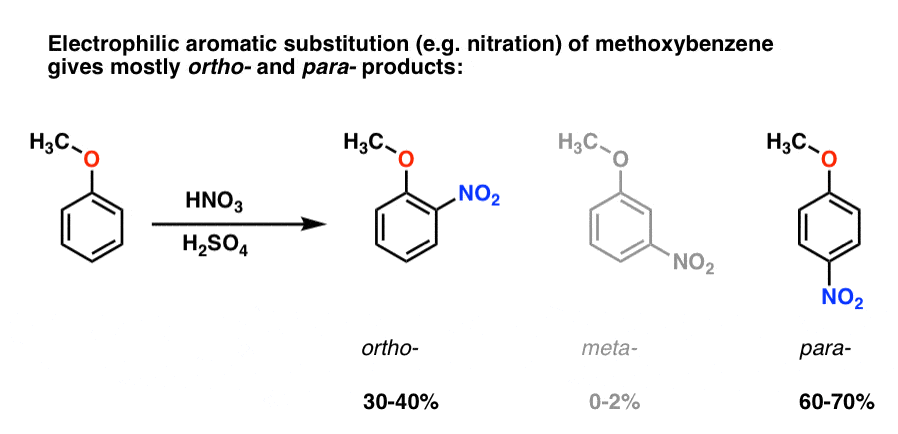
Direct steric hindrance and steric acceleration both have been found with suitably placed large substituents and reagents, and indirect effects arising because one group interferes with the orienting power of another also are known. Steric (spatial) effects generally are smaller than electronic effects in determining the characteristics of reaction by this mechanism, but they are not negligible. These effects are very specific with regard to the position at which the modifying group is introduced. As would be expected, substituents that release electrons toward the reaction site facilitate the reaction, and those that withdraw electrons retard reaction. Reactivity by this mechanism is dominated by the electrophilic character of the reagent (E) however, it also responds powerfully to changes in structure of the organic substrate. 
Examples are known in which the removal of the proton from the carbonium ion intermediate (to form the second pi complex) becomes rate-determining. This complex forms rapidly in an equilibrium preceding the rate-determining step, which itself leads to a carbonium ion intermediate and then by way of a second pi complex to the product. Here, Y represents a substituent on the ring the arrow from the ring centre indicates coordination.Īs shown, the reaction begins with formation of a pi complex, in which the electrons associated with the aromatic ring, or other unsaturated centres (pi electrons), coordinate weakly with the electrophile. Both substituents can be any one of various groups (e.g., hydrogen atoms or nitro, bromo, or tert-alkyl groups). One of the more common is shown here reactions in this category consist of replacement of a group designated Y (often a hydrogen atom) in an aromatic molecule by an electrophilic agent designated E. Reaction by any one of several mechanisms is possible.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
